|
This report also briefly summarizes American Association for the Study of Liver Diseases (AASLD) guidelines for maternal antiviral therapy to reduce perinatal HBV transmission, published previously ( 5). removal of permissive language for delaying the birth dose until after hospital discharge.vaccination for persons with chronic liver disease (including, but not limited to, those with hepatitis C virus infection, cirrhosis, fatty liver disease, alcoholic liver disease, autoimmune hepatitis, and an alanine aminotransferase or aspartate aminotransferase level greater than twice the upper limit of normal) and.single-dose revaccination for infants born to HBsAg-positive women not responding to the initial vaccine series.postvaccination serologic testing for infants whose mother’s HBsAg status remains unknown indefinitely (e.g., when a parent or person with lawful custody surrenders an infant confidentially shortly after birth).testing HBsAg-positive pregnant women for hepatitis B virus deoxyribonucleic acid (HBV DNA).universal hepatitis B (HepB) vaccination within 24 hours of birth for medically stable infants weighing ≥2,000 grams.The following recommendations are new or updated: This report also briefly summarizes previously published American Association for the Study of Liver Diseasest guidelines for maternal antiviral therapy to reduce perinatal HBV transmission. These recommendations also provide CDC guidance for postexposure prophylaxis following occupational and other exposures. ACIP recommends vaccination of adults at risk for HBV infection, including universal vaccination of adults in settings in which a high proportion have risk factors for HBV infection and vaccination of adults requesting protection from HBV without acknowledgment of a specific risk factor. ACIP recommends testing all pregnant women for hepatitis B surface antigen (HBsAg), and testing HBsAg-positive pregnant women for hepatitis B virus deoxyribonucleic acid (HBV DNA) administration of HepB vaccine and hepatitis B immune globulin (HBIG) for infants born to HBV-infected women within 12 hours of birth, followed by completion of the vaccine series and postvaccination serologic testing universal hepatitis B vaccination within 24 hours of birth, followed by completion of the vaccine series and vaccination of children and adolescents aged <19 years who have not been vaccinated previously. 
This report updates and summarizes previously published recommendations from the Advisory Committee on Immunization Practices (ACIP) and CDC regarding the prevention of HBV infection in the United States. Persons with chronic HBV infection are at increased risk for cirrhosis and liver cancer and require medical care. 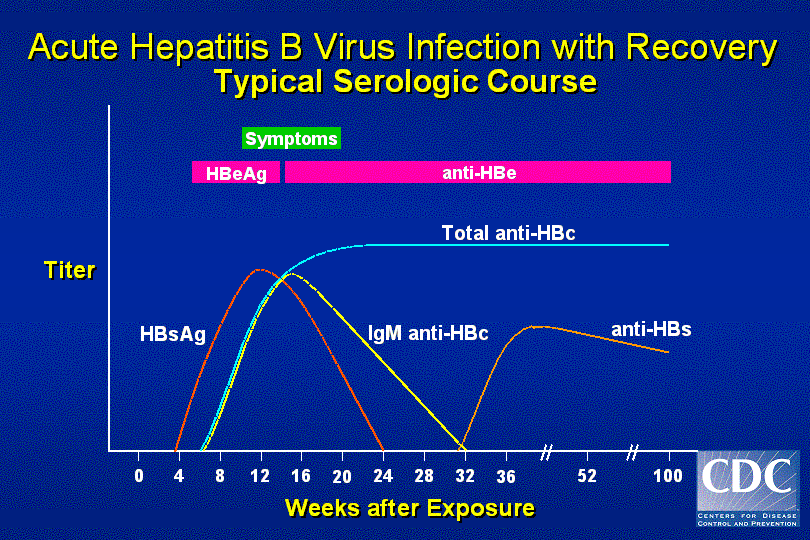
Hepatitis B virus (HBV) is transmitted via blood or sexual contact.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
